Enantioselective liquid chromatography method for the simultaneous determination of chiral and achiral fungicides in aqueous matrices
DOI:
https://doi.org/10.48797/sl.2025.314Keywords:
PosterAbstract
Background: Fungicides are organic compounds, many of them chiral, used in various applications such as medicines, personal care products, agrochemicals and industry. Although they have beneficial effects in controlling fungal plagues and treating diseases, their widespread use has led to their detection in the environment [1-3]. As a result, fungicide contamination is increasingly a global environmental concern, due to the risks it poses to non-target organisms and to human health. Given that enantiomers of chiral fungicides can show different bioactivity, toxicity and degradation, monitoring the enantioselective occurrence of these compounds in the environment is very important to assess the toxicity and adverse effects of each enantiomeric form [3-5]. Objective: The aim of this study is to develop and validate an enantioselective chromatographic method for analyzing a group of five chiral fungicides (ipconazole, metconazole, penconazole, tebuconazole, and tetraconazole) as well as an achiral one (fluconazole) in aqueous matrices. Methods: The chromatographic method was developed using the chiral polysaccharide column Lux i-cellulose 5 [cellulose tris(3,5-dichlorophenylcarbamate)] under reversed elution mode in a liquid chromatograph with a diode array detector. Different compositions and proportions of the mobile phase, various column temperatures and different flow rates were tested. Results: An enantioselective chromatographic method was optimized, allowing to enantioseparate the enantiomers of ipconazole, metconazole, penconazole, tebuconazole, and tetraconazole and of the achiral fungicide fluconazole. Conclusions: This optimized method will be validated and used for the determination of the enantiomers of each chiral target fungicide – ipconazole, metconazole, penconazole, tebuconazole, and tetraconazole – as well as the achiral fungicide fluconazole in water matrices.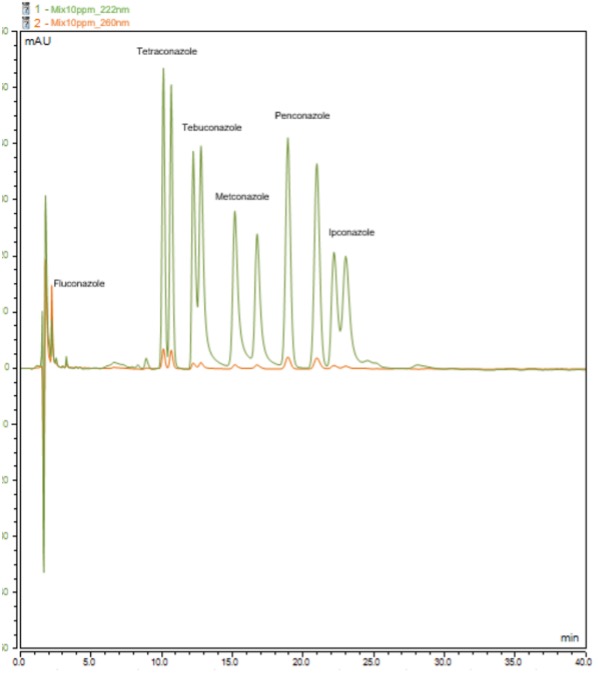
Figure 1. Chromatogram of a mixture of target fungicides, each at 10 mg L-1, at two different wavelengths (222 nm and 260 nm).
References
1. Wang, Y. et al. Occurrence, spatial variation, seasonal difference, and risk assessment of neonicotinoid insecticides, selected agriculture fungicides, and their transformation products in the Yangtze River, China: From the upper to lower reaches. Water Res 2023, 247, 120724, doi: 10.1016/j.watres.2023.120724
2. Ji, C. et al. Enantioselectivity in the toxicological effects of chiral pesticides: A review. Sci Total Environ 2023, 857, 159656, doi: 10.1016/j.scitotenv.2022.159656
3. Zhu, J. et al. Occurrence, spatiotemporal dynamics, and ecological risk of fungicides in a reservoir-regulated basin. Environ Int 2023, 171, 107697, doi: 10.1016/j.envint.2022.107697
4. Draskau, M.K. et al. Azole fungicides and their endocrine disrupting properties: Perspectives on sex hormone-dependent reproductive development. Front Toxicol 2022, 4, 883254, doi: 10.3389/ftox.2022.883254
5. Ribeiro, C. et al. Occurrence of chiral bioactive compounds in the aquatic environment: A review. Symmetry 2017, 9, 215, doi: doi:10.3390/sym9100215
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Beatriz Suordem, Joaquín Marrero, Ana Gorito, Cláudia Ribeiro, Ana Rita L. Ribeiro

This work is licensed under a Creative Commons Attribution 4.0 International License.
In Scientific Letters, articles are published under a CC-BY license (Creative Commons Attribution 4.0 International License), the most open license available. The users can share (copy and redistribute the material in any medium or format) and adapt (remix, transform, and build upon the material for any purpose, even commercially), as long as they give appropriate credit, provide a link to the license, and indicate if changes were made (read the full text of the license terms and conditions of use).
The author is the owner of the copyright.









