In silico investigation of the interaction between paclitaxel and ergosterol in P-glycoprotein
DOI:
https://doi.org/10.48797/sl.2026.465Keywords:
PosterAbstract
Background: Multidrug resistance (MDR) mediated by the overexpression of P-glycoprotein (P-gp) is one of the main obstacles in cancer treatment, as this transporter reduces the intracellular concentration of chemotherapeutic agents such as paclitaxel (PTX) [1]. Natural compounds have shown promise as P-gp modulators for reversing MDR [2]. Ergosterol (ERG), a sterol commonly found in fungi, has shown potential as a modulator of this transporter [3]. However, the molecular details of its interaction and its combination with PTX are not yet fully understood. Objective: To investigate the molecular interactions between PTX and ERG at the P-gp binding site using single molecular and sequential docking simulations to assess possible competitive effects. Methods: The human P-gp structure (PDB: 7A6E) and the AutoDock VINA algorithm were used. Individual docking studies were performed to characterize the isolated affinity of each ligand. Subsequently, a sequential docking strategy was applied, in which one ligand was held fixed in the best conformation, and the second was docked again at the same site, simulating prior occupation of the transporter. Results:In the individual binding assays, both PTX and ERG exhibited high affinity, with binding energies of –10.77 kcal/mol (Kd ≈ 12.60 nM) and –10.58 kcal/mol (Kd ≈ 17.30 nM), respectively. In sequential docking, the prior presence of ERG significantly reduces the affinity of PTX (-8.04 kcal/mol), resulting in an increased dissociation constant in the micromolar range (12.70 µM). Conversely, ERG maintained a favorable affinity (-10.19 kcal/mol) even in the presence of PTX. Ligand efficiency (LE) analysis revealed that ERG (LE = 0.365) exhibits superior energy utilization to PTX (LE = 0.174) relative to its molecular size. Conclusions: Structural analysis reveals that ergosterol exerts steric interference at the P-gp binding site, resulting in partial competition with paclitaxel. Due to its higher binding affinity, ergosterol partially obstructs the transporter’s binding pocket, which may impair the binding stability and transport efficiency of the chemotherapeutic agent without preventing its initial interaction with the active site.
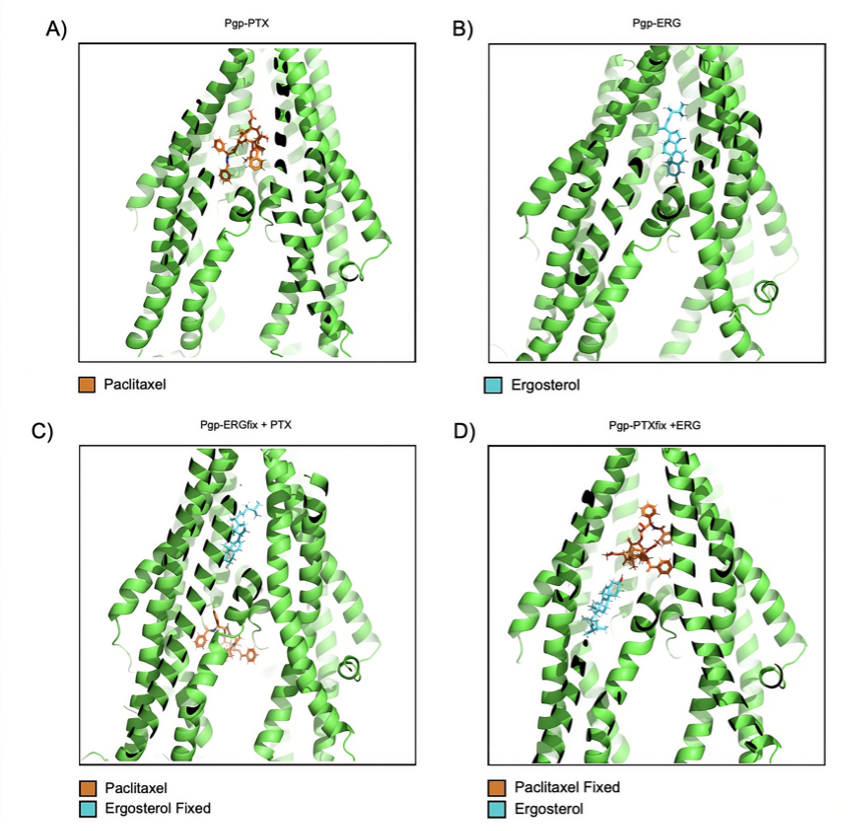
Figure 1. In silico representation of the co-occupation and structural overlap of paclitaxel and ergosterol in the binding pocket of human P-gp (PDB: 7A6E).
References
1. Dong, J. et al. Strategies to overcome cancer multidrug resistance (MDR) through targeting P-glycoprotein (ABCB1): An updated review. Pharmacol Ther 2023, 249, 108488, doi:10.1016/j.pharmthera.2023.108488.
2. Fonseca, J. et al. The Potential of Mushroom Extracts to Improve Chemotherapy Efficacy in Cancer Cells: A Systematic Review. Cells 2024, 13(6), 510, doi:10.3390/cells13060510.
3. Rangsinth, P. et al. Potential Beneficial Effects and Pharmacological Properties of Ergosterol, a Common Bioactive Compound in Edible Mushrooms. Foods 2023, 12, doi:10.3390/foods12132529.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Jéssica Fonseca, Josiana Vaz, Sara Ricardo

This work is licensed under a Creative Commons Attribution 4.0 International License.
In Scientific Letters, articles are published under a CC-BY license (Creative Commons Attribution 4.0 International License), the most open license available. The users can share (copy and redistribute the material in any medium or format) and adapt (remix, transform, and build upon the material for any purpose, even commercially), as long as they give appropriate credit, provide a link to the license, and indicate if changes were made (read the full text of the license terms and conditions of use).
The author is the owner of the copyright.









